Some people may reduce their need for pain medications entirely with SCS therapy. Individual results will vary.
Is SCS right for me?
SCS therapy might be a good fit for you if you have:
- Chronic pain that’s lasted six months or more
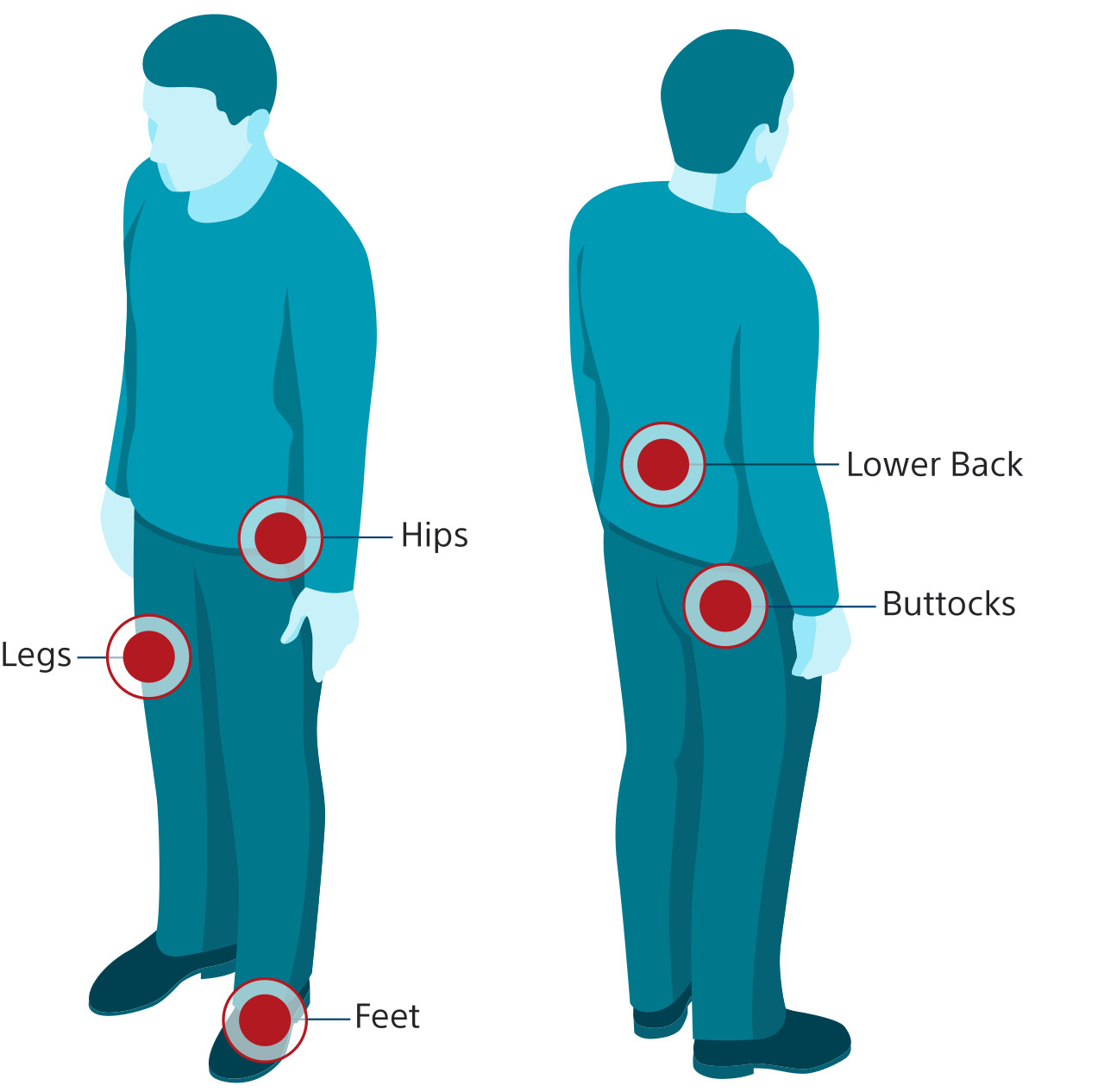
- Pain in your back, legs, feet, or a combination of those areas due to:
- Failed Back Surgery Syndrome (FBSS)
- Painful Diabetic Peripheral Neuropathy (DPN) of the lower extremities
- Complex Regional Pain Syndrome (CRPS)
- Radicular Pain Syndrome
- Other neuropathic pain conditions
A pain management specialist can tell you if SCS therapy may be right for you.
See the complete SCS indications and contraindications.
Have you been suffering from pain for more than 6 months?
How does SCS work?
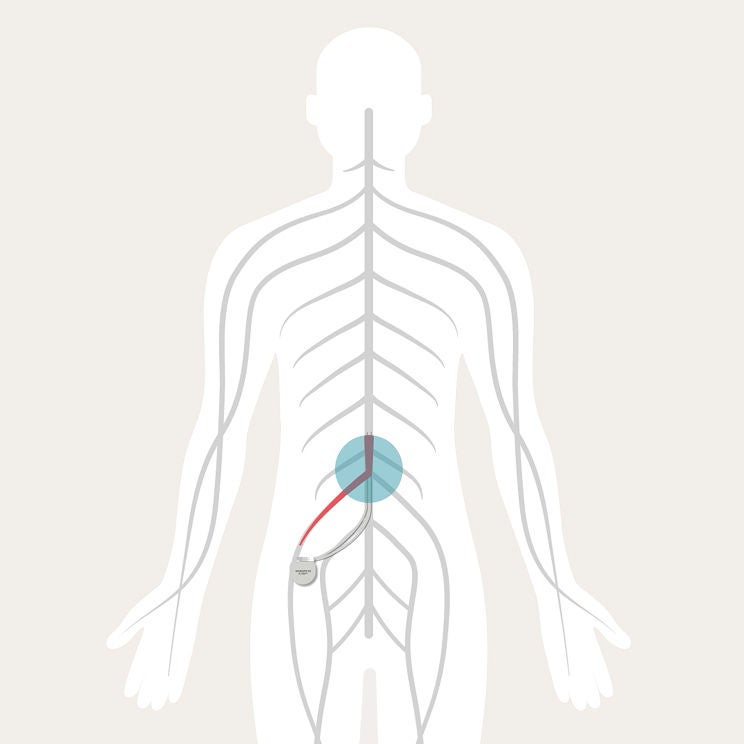
You feel pain when your nerves send pain signals to your brain through your spinal cord. Your SCS System is designed to interrupt those signals, so you don’t feel them as pain.
The SCS System is made up of a stimulator and leads, which are placed in your back. The stimulator sends mild pulses through the leads. These pulses interrupt pain signals on their way to your brain.
See how SCS works
Watch this short, animated video to find out more about how SCS works and how it might help you find relief.
See the proof
Clinical studies and trials have shown that SCS is effective in relieving chronic pain.
100K+ people treated
Over 100,000 people across the globe have found pain relief from Boston Scientific SCS therapy. Relief that’s long-term, drug-free, FDA-approved, and effective.
90% success rate in trials
More than 90% of Boston Scientific SCS trial participants reported a successful experience and decided to get the permanent implant.1
95% would recommend
9 out of 10 Boston Scientific SCS patients said they were satisfied with their therapy. 95% would recommend it to others.1
What to expect from Spinal Cord Stimulation (SCS)
SCS is designed to help you get long-term pain relief in different parts of your body. You can test drive SCS in a short trial. If your trial is successful, your pain management specialist can help you get a permanent implant.


Discover the WaveWriter AlphaTM SCS System
Boston Scientific’s SCS Systems offer:
- Relief you feel in minutes — that lasts for years
- Therapy that can be personalized to your unique pain
- Therapy proven to get you back to everyday activities
Getting you back to everyday life
The goal of SCS therapy is to get you back to doing the everyday things you enjoy. How? With long term relief you can rely on.
Need proof? One major clinical study showed that patients using Boston Scientific’s SCS therapies reported a greater improvement in their ability to do everyday activities† after two years than patients using non-Boston Scientific SCS Systems after three months.2, 3, 4 That means lasting, long-term improvement.

Try SCS first
Take SCS for a short “test-drive” before you commit. You can try a temporary trial system for three to seven days to see if SCS is right for you. If your results are successful, your doctor can help you move forward with a permanent implant.
The mySCSTM App
The app lets you track your activity and pain levels, connect with your Boston Scientific care team, and learn more about what to expect along the way.
Significant long-term relief
Another major clinical study showed great results from SCS. Patients with severe low-back pain (8 or above on a 0-10 pain scale) reported a nearly 6-point drop in pain score after two years.5
- Neil
- Karen
- Mary
The patient quotes in this material describe real personal experiences. Individual results may vary. Patients can experience different levels of pain management and different changes in their activities and use of medications. Consult with your physician to determine if you are a candidate for this procedure and what you may gain from the therapy.
Frequently asked questions
Yes. Even though your leads and stimulator are surgically implanted, your doctor can disengage or remove them as needed.
Not all devices are MRI conditional, but the WaveWriter AlphaTM Systems allow for access to full-body MRI scans under specified conditions.
The stimulator is about the size of a silver dollar and placed in a spot that’s comfortable for you. You might feel it if it's pressed on, but most people don’t notice it, and others can’t see it.
Yes. A key benefit of SCS is that you can adjust your therapy settings to respond to changes in your pain or switch between programs if you prefer different therapies during different activities.
The mySCS GO™ Therapy Controller App* is designed to make controlling your Boston Scientific SCS System quick and simple. The innovative app comes already loaded on an Android smart device, and provides:
- Greater personalization of your therapy
- The ability to switch between programs quickly and easily
- The ability to adjust the level of your stimulation
- User-friendly, touchscreen controls
You decide on a recharging schedule that best fits your needs. Most people choose to charge based on a routine that fits their lifestyle, like during their favorite weekly TV show. Your wireless remote control will also tell you when your implant battery is low and needs recharging.
Recharging the stimulator battery is designed to be very simple. The charger is lightweight, wireless, and portable, so you can even charge on the go. During charging, you use a soft cloth belt (included with the charger) to help place and keep the charger over the device.
SCS is covered by most health insurance plans, including Medicare, commercial insurance, and workers' compensation. Your doctor will need to secure preauthorization by providing your insurer with the required documentation. For specific details about coverage, benefits, and out-of-pocket costs, consult your doctor’s office or your insurance provider directly.
SCS resources
Find the tools you need to take your next step. Order or download information kits, guides and brochures — including our Doctor Discussion Guide — videos, and more.
Take your next step toward pain relief
Have you been suffering from pain for more than 6 months?
References
- Thomson SJ, Kruglov D, Duarte RV. A spinal cord stimulation service review from a single centre using a single manufacturer over a 7.5 year follow-up period. Neuromodulation. 2017;20(6):589-599. N=321
- Clark S. Metzger, M. Blake Hammond, Jose F. Paz-Solis, William J. Newton, Simon J. Thomson, Yu Pei, Roshini Jain, Michael Moffitt, Luca Annecchino & Que Doan (2021) A novel fast-acting sub-perception spinal cord stimulation therapy enables rapid onset of analgesia in patients with chronic pain, Expert Review of Medical Devices, DOI: 10.1080/17434440.2021.1890580. (N=41)
- FAST MOA computational modeling by Dr. Warren Grill’s lab at Duke University. Gilbert et al., Computational modeling predicts dorsal columns are involved in fast-acting sub-perception spinal cord stimulation (SCS). SFN 2021.
- Wallace MS, North JM, Phillips GM, Calodney AK, Scowcroft JA, Popat-Lewis BU, Lee JM, Washabaugh EP 3rd, Paez J, Bolash RB, Noles J, Atallah J, Shah B, Ahadian FM, Trainor DM, Chen L, Jain R. Combination therapy with simultaneous delivery of spinal cord stimulation modalities: COMBO randomized controlled trial. Pain Manag. 2023 Mar;13(3):171-184.
- Kapural, Cong Yu, et al. Novel 10-kHz High-frequency Therapy (HF10 Therapy) Is Superior to Traditional Low-frequency Spinal Cord Stimulation 90for the Treatment of Chronic Back and Leg Pain: The SENZA-RCT Randomized Controlled Trial. Anesthesiology 2015; 123:851 -860
*For a list of compatible devices, visit www.myscsjourney.com/mySCSGO